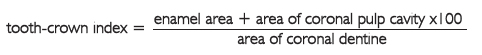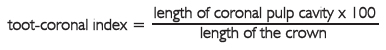The Coronal Pulp Cavity Index: A Forensic Tool for Age Determination in Human Adults
Índice de la cavidad coronal pulpar: una herramienta forense para la determinación de la edad en humanos adultos
Cuad Med Forense. 2008; 14(53-54):235-249
Fecha de recepción: 9.DIC.08; Fecha de aceptación tras revisión interna: 29.DIC.08
ABSTRACT
The correlation between the reduction of the coronal pulp cavity and the chronological age was examined in a sample of 846 intact teeth from 433 individuals of known age and sex. Panoramic (rotational) radiography was used to measure the length (mm) of the tooth crown (CL=coronal length) and the length (mm) of the coronal pulp cavity (CPCL=coronal pulp cavity length). The tooth-coronal index (TCI) after Ikeda et al. (1985) was computed for each tooth and regressed on the real age of the sample. The correlation coefficients ranged from -0.92 (molars, combined sample, right side) to -0.87 (female molars), with a S.E. of the estimate ranging from 5.88 to 6.66 years. Correlations were slightly higher in males than females. The obtained equations allowed estimation of age in a sample of 100 teeth of both sexes (not used for the regression) with an error of ± 5 years in 81.4% of cases for the male molars. This study shows the potential value of a little-known aging method, which can be easily used to estimate age both in living individuals and skeletal material of unknown age in a forensic context.
Key words: Coronal pulp cavity index, age, adults, forensic odontology.
RESUMEN
La correlación entre la reducción de la cavidad coronal pulpar y la edad cronológica ha sido estudiada en una muestra de 846 dientes intactos procedentes de 433 individuos de sexo y edad conocidos. La radiografía panorámica ha sido utilizada para medir el largo (mm) de la corona (CL=coronal length) y el largo (mm) de la cavidad coronal pulpar. El índice dentario Tooth coronal index (TCI) según Ikeda et al. (1985) ha sido calculado por cada diente para determinar la edad real del individuo con el método de la regresión. Los coeficientes de regresión varían de -0.92 (molares, muestra total, lado derecho) a -0.87 (molares femeninos) con un S. E. de estimación de 5.88 a 6.66 años. Las correlaciones son más altas en los varones respecto a las mujeres. Las ecuaciones obtenidas lograron estimar la edad en una muestra de 100 dientes (no utilizados en la regresión) con un error de ± 5 años en el 81.4% de los casos por los molares masculinos. El estudio ilustra el valor potencial de un método que puede ser utilizado para estimar la edad en individuos vivos y material esquelético de edad desconocida.
Palabras clave: Índice coronal pulpar, edad, adultos, odontología forense.
Introduction
Among the numerous methods of age estimation on the basis of teeth, the progressive modification of the coronal pulp cavity has received far less research attention than, for example, has attrition (Brothwell, 1989; Benfer and Edwards, 1991; Lovejoy et al., 1985; Miles, 1958, 1963; Nowell, 1978; Smith, 1984; Solheim, 1988; Takei, 1984; Tomaru et al., 1993; Walker et al., 1991; Wei and Feng, 1984) and root dentine transparency (Bang, 1989; Bang and Ramm, 1970; Burns and Maples, 1976; Drusini, 1991; Drusini et al., 1991; Johanson, 1971; Lamendin and Cambray, 1981; Lamendin et al., 1990; Lamendin et al., 1992; Pilz, 1959; Solheim and Sundnes, 1980; Solheim, 1989, 1993). Other promising methods of age determination from teeth concern cementum annulation (Charles et al., 1986; Condon et al., 1986; Grobskopf, 1990; Lipsinic et al.,1986; Lorton, 1988; Naylor et al., 1985; Stott et al., 1982), counting of cross-striations and Striae of Retzius in juveniles (Huda and Bowman, 1994 and 1995), and aspartic acid racemization (Helfman and Bada, 1975, 1976; Masters, 1986; Ogino et al., 1985; Ogino and Ogino, 1988; Ohtani and Yamamoto, 1987, 1991, 1992; Ritz et al., 1990, 1993). Fluorescence from dentine and cementum (Kvaal and Solheim, 1989) still need more investigation.
After a review of the literature, Rösing et al. (1998) stated that the best method of age determination from teeth is the cement ring count, both for reliability and for general applicability. A close competitor is racemization, which is roughly comparable in accuracy, but not in applicability, and is best suited for fresh corpses; moreover, the method is influenced by the temperature conditions during the time after death. As a matter of fact, both methods are quite time expensive and require some sophisticated laboratory equipment. Then, other easy methods are to be explored for practice in historic and prehistoric anthropology as well as forensic osteology (Schmidt, 2008); recently, Harris (2008) published a paper on statistical applications in dental anthropology, and Liversidge (2008) and Schmidt (2008) revisited the dental age estimation methods.
Since 1925 Bodecker identified the apposition of secondary dentine as being related to chronological age. Detailed studies of the pattern and rate of secondary dentine apposition in upper anterior teeth were performed by Philippas and Applebaum (1966, 1967, 1968), but without the goal of estimating age at death. The secondary dentine deposition was included in the method pioneered by Gustafsons (1950), where the dentine transparency and the secondary dentine values showed the highest correlation with age, as the following studies of Johanson (1971), Maples (1978), and Metzger et al. (1980) have demonstrated. Other authors, as Nalbandian and Sognnaes (1960), were also of the opinion that it was necessary to include the secondary dentine for estimating age. Some authors on the contrary argued that secondary dentine changes have not proven useful as a biomarker for systemic aging (Lamendin et al., 1990; Morse, 1991).
Secondary dentine begins to form once the tooth crown is fully formed, the tooth is in occlusion and the root complete (Costa, 1986). Dentine formation does continue throughout life, and layers of secondary dentine (also known as regular secondary dentine: Berkovitz et al., 1992) are continuously deposited by odontoblasts lining the pulp chamber. Since regular secondary dentine is laid down at the pulpal end of the primary dentine, the pulp cavity decreases in size with age (Gustafson, 1950; Balogh, 1957; Philippas, 1961; Johanson, 1971; Hillson, 1986; Morse, 1991; Berkovitz et al., 1992; Solheim, 1992). The pattern for the secondary dentine deposition varies among different teeth: for example, in molars the greatest dentine deposition is on the floor of the pulp chamber, and lesser amounts are deposited on the occlusal and lateral walls (Philippas and Applebaum, 1967, 1968; Morse, 1991). As regular secondary dentine is deposited in larger amounts on the floor of the pulp chamber than on the roof, some authors suggested that age has a greater influence than does attrition or irritation on secondary dentine formation (Philippas and Applebaum, 1966; Berkovitz et al., 1992). As a matter of fact, changes in the structure of dentine may be also related to noxious or pathological stimuli, which include caries, traumatic occlusion and temperature extremes (Bang, 1989). Where dentine is subjected to acute damage, as for example dental caries, some of the underlying odontoblasts die, while others lay down a repair tissue. This irritation or response dentine is often known as irregular secondary dentine, and sometimes referred to as tertiary dentine (Berkovitz et al., 1992).
Therefore, there are two types of secondary dentine, one of them is laid down continuously with increasing age (regular secondary dentine), and the other (irregular or tertiary dentine) is dependent on pathological conditions (Robinson and Boling, 1952; Philippas, 1961; Johanson, 1971; Berkovitz et al., 1992). Regular secondary dentine usually contains fewer dentinal tubules dispersed regularly through it and may be difficult to distinguish from the primary dentine. On the other hand, to distinguish the two types of secondary dentine is relatively easy: in the irregular secondary dentine the tubules are few in number, as in regular secondary dentine, but they are irregularly arranged (Hillson, 1986; Berkovitz et al., 1992). Worn and unworn teeth from the same mouth tend to show the same amount of regular secondary dentine (Burns and Maples, 1976). Occasionally, small isolated bodies of dentine (denticles, Pindborg, 1970; Morse, 1991) form within the pulp chamber.
As the pulp cavity diminishes in size the amount of secondary dentine can be used for age determination. Ito (1972, 1975) took note of the change in the form of the anatomical crown and the constriction of the coronal pulp cavity. Buccolingual sections of a thickness of 1.0 mm were photographed with a Softex, CMR type, with secondary voltage of 90 V, secondary electric current of 5 mA, and an exposure time of 10 seconds. The film used was a Softex Film FG. Ito calculated the areas of the enamel, coronal dentine and coronal pulp cavity of 302 permanent teeth (excluding the third molar), representing the respective area ratio as a tooth-crown index:
Ito (1972, 1975) found a negative correlation between the tooth-crown index and the real age ranged from -0.38 (molars) to -0.63 (anterior teeth), with a standard error of the estimate ranging from 8.6 to 6.7 years. Haertig and Durigon (1978) compared the method of Ito with the method of Gustafson and found than the former was not more precise even if considered more rigorous from the theoretic point of view.
In 1985, Ikeda et al. took X-ray photographs of 116 extracted human teeth (53 incisors and 63 molars), and made their quadruple size prints. On the prints, he measured the lengths of the coronal pulp cavity and crown and calculated the tooth-coronal index:
The index decreased with increasing age, with a very high correlation coefficients: -0.91 for the incisors (S.E. = 6.06), and -0.93 for the molars (S.E. = 5.73). In a previous study Drusini (1993), following in part the Ikeda et al. method (intact teeth instead of sections were used) confirmed the negative correlation between tooth-coronal index and age on 68 premolars and 98 molars: the correlation coefficients ranged from -0.73 (female molars) to -0.89 (female premolars). The aim of this paper is to verify the method of Ikeda et al. (1985) with a higher number of teeth from individuals of known sex and age and to test the regression equations on a sample of teeth not used for the regression. As a matter of fact, neither Ito nor Ikeda et al. have tested the method for age estimation on a control sample.
Beginning in 1991, a research based on a total sample of 1806 human teeth of known sex and age belonging to 1304 individuals of both sexes was carried out in the laboratory of physical and forensic anthropology of the University of Padua (Italy). The research mainly concerned two methods for age determination in unknown human bodies and skeletons. The correlation between the amount of root dentine transparency (RDT) and chronological age was first examined in a sample of 366 intact teeth from 295 individuals of known sex and age. Correlations, especially for anterior teeth (incisors and canines) were slightly higher in females than males, even if neither the slopes nor the Y intercepts of the regression lines were significantly different. Linear regression analysis to predict age separately for males, females and the total sample resulted in standard errors of estimate ranging from 8.88 to 11.46 years (Drusini, 1991; Drusini et al., 2000). RDT was measured by two techniques: 1) computerized densitometric analysis and 2) vernier caliper. Age estimations based on computerized densitometric analysis were no more accurate than were those determined by caliper measurement: both give a predictive success of ± 5 years in about 45-48% of cases for premolars. The television-based digitization system has some disadvantages: it is expensive, not portable, and requires some training to be used. However, it furnishes a more standardized method, a rapid graphic illustration of the results, and an immediate storage of statistical information for future use (Drusini et al., 1991).
In 1993, soft X-ray photos of 68 premolars and 98 molars from 102 males and 64 females were taken to measure the length (mm) of the crown (CL) and the length of coronal pulp cavity (CPCL). Then, the coronal index CI=CPCLX100/CL after Ikeda et al. (1985) was computed for each toot and regressed on the actual age of the individuals to obtain equations that allow estimation of age in unknown bodies and isolated teeth (Drusini, 1993). The correlation coefficients ranges from -0.73 (female molars) to -0.89 (female premolars). No significant differences were observed in age estimation using sex-specific equations.
Material and methods
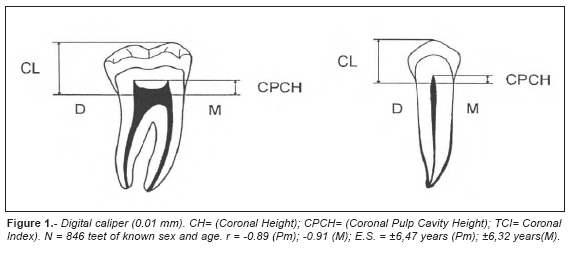
The sample consisted of panoramic X-ray photos of 846 intact teeth (425 premolars and 421 molars) from 433 Caucasian individuals (213 males and 220 females) of known sex and age from Veneto region of Northern Italy. The age of the individuals ranged from 9 to 76 years with a mean age of 34.68 for the males, and 34.33 for the females. Panoramic X-ray photographs were provided by private radiologists, dentists and hospitals. As the panoramic (rotational) radiography technique is higly standardized, teeth coming from various laboratories does not represent a problem for the interpretation. In essence, the panoramic radiography is a tomography of a curved layer, with the X-ray tube and the film rotating around the patient (Langland et al., 1982; Poyton, 1982). The advantages of using panoramic X-ray photographs, compared to the Ikeda et al. (1985) method, are: 1) cutting the teeth is not necessary; 2) panoramic X-ray is a very common dental-test, so a large casuistry can be examined in a short time; 3) all the mandibular and the maxillary teeth are on one film; 3) gross lesions (fractures, unerupted teeth) are readily visible. Panoramic X-ray show a lack of detail in comparison with the intraoral radiography, especially for the anterior teeth, and the projection can only be taken at one angle. On the basis of these remarks, we decided to consider only the posterior teeth where the pulp chamber is normally fully visible, disregarding all the pathological specimens.
For each radiograph we considered mandibular premolars and molars (excluding the third) of the mandible only, because mandibular teeth are more visible than the maxillary ones. We chose the side where the pulp chamber was more visible. The following measurements (in mm) were taken independently on the photographs by two observers, using a digital caliper to the nearest 0.01 mm: length of the crown (CL), and length of the coronal pulp cavity (CPCL). Then, with the mean of the two measurements, the tooth-coronal index (TCI) for each tooth was calculated as follows (Fig. 1):
The use of an index obviate the need to standardize the tooth size on the photographs. We divided the teeth in two groups: premolars and molars, for the statistical analysis. Simple linear regression using STATISTICATM package of the Statsoft, Inc. was carried out by regressing the proportional coronal pulp cavity length (TCI) on the actual age for each group of teeth, for males and females, and for the combined sample. We used 375 premolars (178 male and 197 female) and 371 molars (182 male and 189 female). We calculated the TCI also in young individuals (under 20) in order to fix the upper limit of the CPCL. Finally, 100 teeth not included in the regression analysis were used as a test sample.
Results
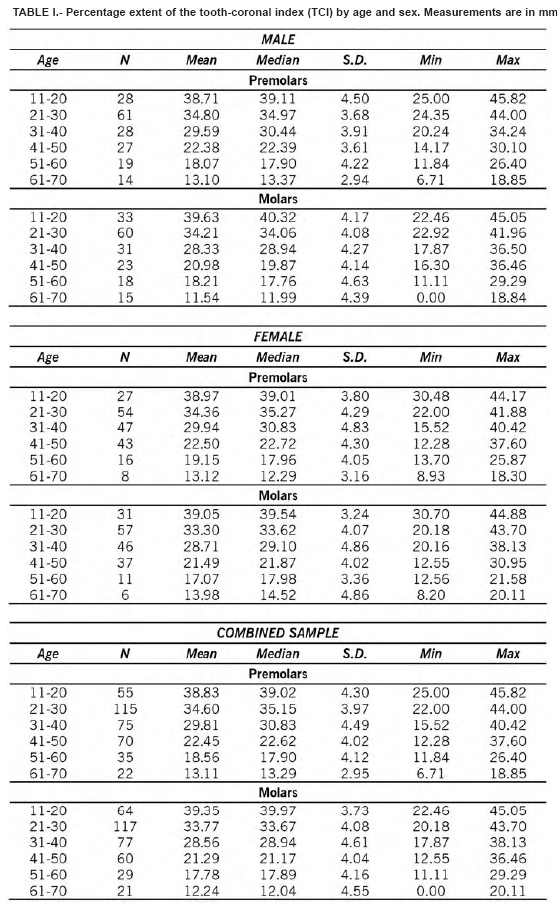
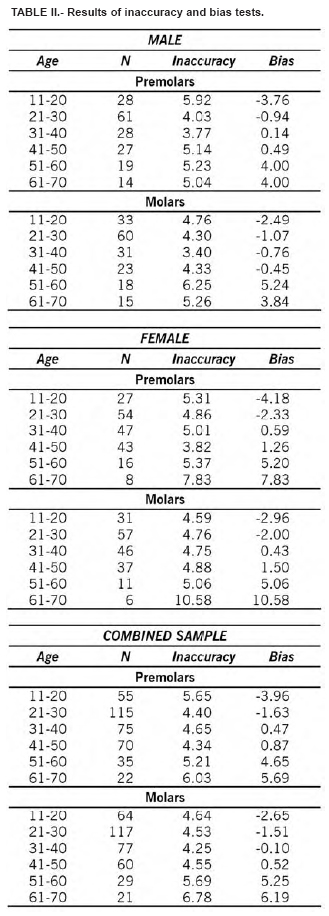
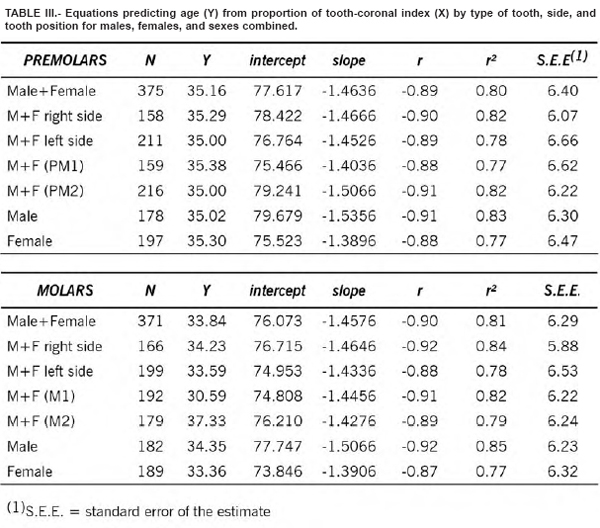
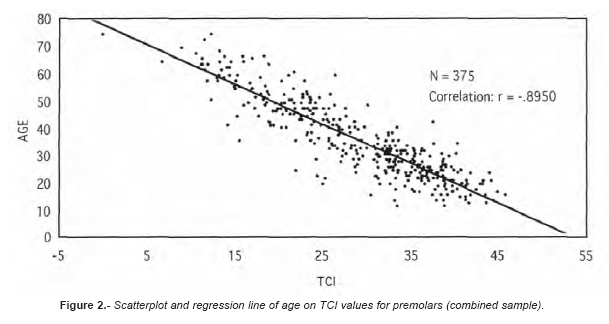
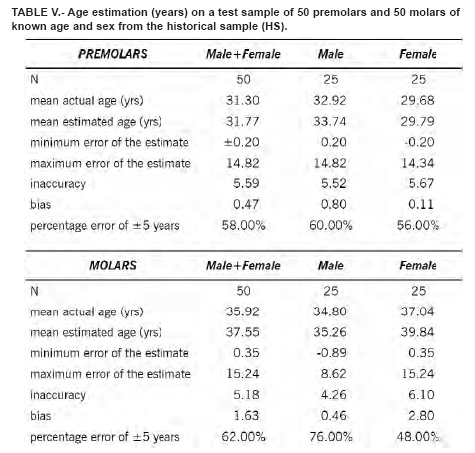
Table 1 shows the extent of the TCI by age category. Replicate measurements were nearly identical, with an inter-observer error of 3.8%. Means, medians, ranges, and standard deviations for males, females, and the combined sample are also compared. As suggested by Lovejoy et al. (1985), the inaccuracy (mean absolute error) and bias (mean error) for each decade in the three samples for the two types of teeth are also provided (Table II). These tests are necessary since the correlation coefficient as a measure of association is influenced by the age range and composition of the test sample which normally differs for each analysis. The simple linear regression results are shown in Table III. The correlations were significant especially for molars in males (r = -0.92, r2 = 0.85). The deviations, i.e. true age minus the predicted value of the regression line, were slightly smaller for males. Figure 2 shows the scatterplots and the regression lines of age (Y) and the TCI (X) values for premolars (combined sample).
The regression equations are as follows:
Premolars
- Y = 77.617 – 1.4636X combined sample
- Y = 79.679 – 1.5356X males
- Y = 75.523 – 1.3896X females
Molars
- Y = 76.073 – 1.4576X combined sample
- Y = 77.747 – 1.5066X males
- Y = 73.846 – 1.3906X females
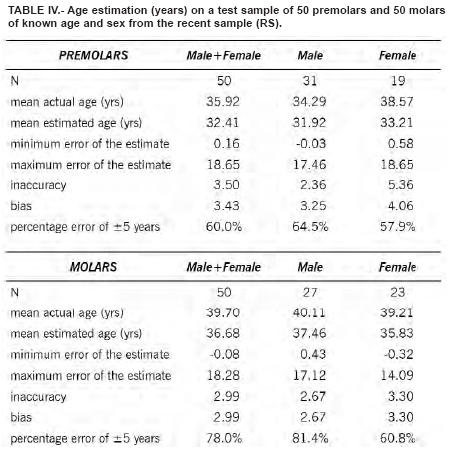
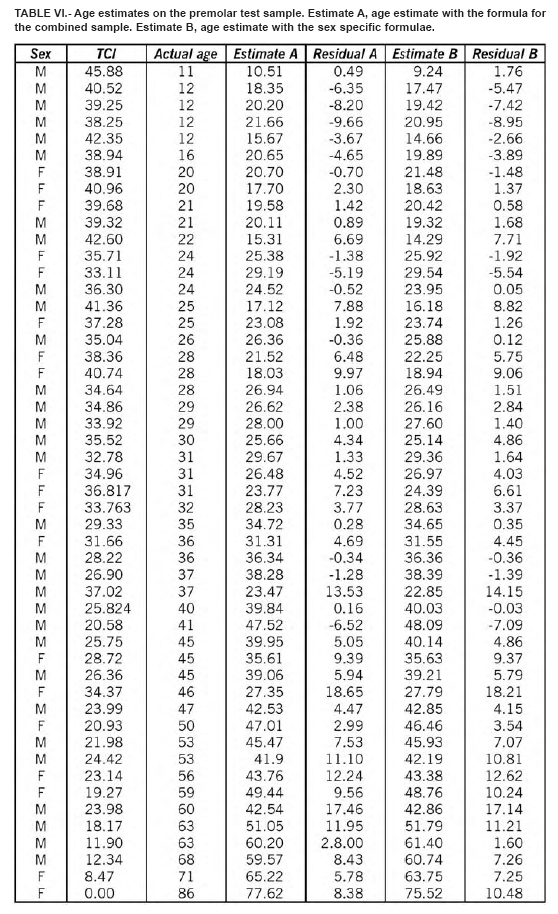
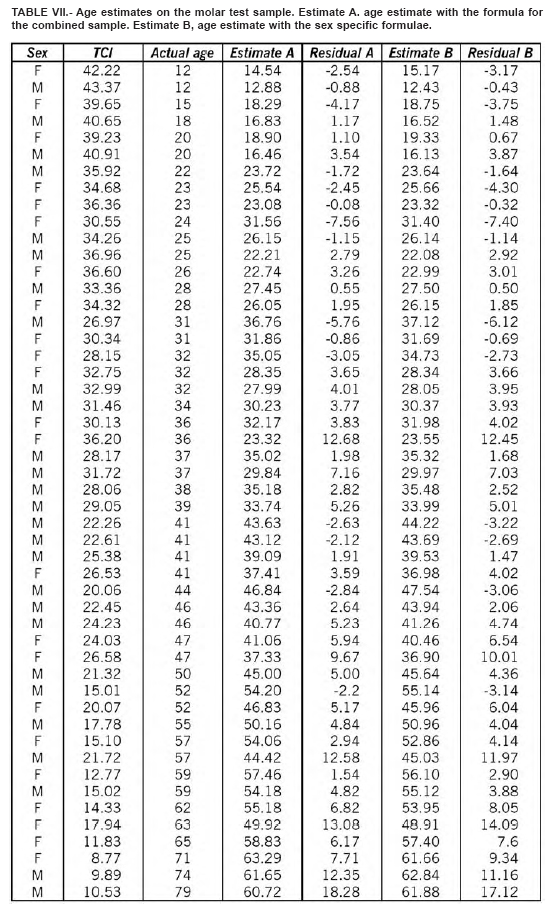
The regression equations were tested on a random sample of 100 teeth (50 premolars and 50 molars) not used for the regression analysis. Three equations were used: one for the males, one for the females and one for the sexes combined. Results are shown in Table IV. The best estimation were obtained for the male molars, with an error of ± 5 years in the 81.4% of the cases. No significant difference do exists between the teeth of the right and the left side, and between first and second premolar and first and second molar.
Discussion and conclusion
The length of the coronal pulp cavity shows a significant correlation with individual chronological age: the coefficients of correlation we found for molars are practically the same of Ikeda et al. (1985). The better correlations of the molar group may be related to a lower resolution of the panoramic radiography for the premolars, where the pulp chamber upper limits are sometimes less visible. Sex of the individual appears to have no significant influence on age determination using tooth-coronal index, so that sex-specific formulae are not necessary for age determination in specimens of unknown sex. The panoramic technique is no better than the intraoral films, but the possibility to have on one film all the teeth of both dental arches represents undeniably an advantage, so one can also compare the TCI trend of all groups of teeth in the same individual. The index obviates the need to standardize the tooth size on the photographs, so dental X-ray obtained with different techniques can be used. The coefficients of correlation are higher in comparison with the coefficients obtained with the analysis of root dentine transparency on a sample of teeth from the same population (Drusini, 1991; Drusini et al., 1991).
The tooth coronal index is then a reliable biomarker for age assessment in living individuals of unknown personal data for human biology studies, and also in the forensic context. This method may also be applied to estimate age in archaeological specimens using both TCI, tooth wear and other age estimation methods for comparison.
Recently, in collaboration with the Anthropology Department of Lodz (Poland), from age estimation procedures we chose two for comparison: one of them is the standard method based on root dentine transparency (RDT); the other one is based on tooth coronal index (TCI). The material consisted of X-ray photographs and longitudinally sectioned teeth of 276 individuals (174 males and 102 females) of known age. The comparison shows that the poorly known TCI method is at least as precise as the most widely applied and acknowledged procedure based on longitudinal sections of teeth (Zadzinska et al., 2000).
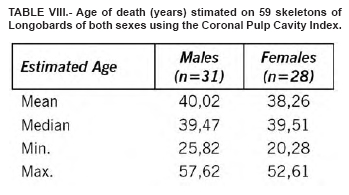
This method may also be applied to estimate age in archaeological specimens using both TCI and tooth wear and other age estimation methods for comparison. We tried to estimate the age at death in a sample of skeletons of both sexes from Longobard age (568 – 756 d.C.), recovered during the Dueville and Sovizzo (North-East Italy) excavations carried out in 1990-1997 and 2000-2002. The estimated age was concordant with the basic age markers of the skeletons currently used by forensic anthropologists (Tables V, VI, VII, VIII).
Acknowledgments
We are deeply indebted to all the people who made available panoramic radiographies for this study: Dr. P. Angi, Dr. R. Bastianello, Dr. F. Coppola, Dr. F. Marcon, Prof. G. Silvestre of the Radiology Service of the Cittadella Hospital, and Prof. S. Vio.
References
- Balogh K (1957) Die Veränderungen der Mundgebilde und Zähne im vorgerückten Alter. Zahnärtzl. Prax. 8 : 2-3.
- Bang G (1989) Age changes in teeth: developmental and regressive. In MY Isçan (ed.): Age Markers in Human Skeleton. Springfield: CC Thomas, pp.211-235.
- Bang G and Ramm e (1970) Determination of age in humans from root dentin transparency. Acta Odont. Scand. 28:3-35.
- Benfer RA, and Edwards DS (1991) The principal axis method for measuring rate and amount of dental attrition: estimating juvenile or adult tooth wear from unaged adult teeth. In MA Kelley and CS Larsen (eds.): Advances in Dental Anthropology. New York: Wiley-Liss, Inc., pp. 325-340.
- Berkovitz BKB, Holland GR, and Moxham BJ (1992) A Colour Atlas and Textbook of Oral Anatomy, Histology and Embryology (second edition). Wolfe Publishing Ltd.
- Bodecker CF (1925) A consideration of some of the changes in the teeth from young to old age. Dental Cosmos 67 : 543-549.
- Brothwell D (1989) The relationship of tooth wear to aging. In MY Isçan (ed.), Age Markers in Human Skeleton. Springfield: CC Thomas, pp. 303-316.
- Burns KR, Maples WR (1976) Estimation of age from individual adult teeth. J. Forensic Sci. 21 : 343-356.
- Charles DK, Condon K, Cheverud JM, and Buikstra JE (1986) Cementum annulation and age determination in Homo sapiens. I. Tooth variability and observer error. Am. J. Phys. Anthropol. 71 : 311-320.
- Condon K., Charles DK, Cheverud JM, and Buikstra JE (1986) Cementum annulation and age determination in Homo sapiens. II. Estimates and accuracy. Am. J. Phys. Anthropol. 71 : 321-330.
- Costa RL Jr (1986) Determination of age at death: dentition analysis. In MR Zimmerman, JL Angel (eds.): Dating and Age Determination of Biological Materials. London: Croom Helm, pp. 248-269.
- Drusini AG (1991) Age-related changes in root transparency of teeth in males and females. Am. J. Hum. Biol. 3 : 629-637.
- Drusini AG, Calliari I, and Volpe A (1991) Root dentine transparency: age determination of human teeth using computerized densitometric analysis. Am. J. Phys. Anthropol. 85 : 25-30.
- Drusini AG (1993) Age estimation from teeth using soft X-ray findings. Anthrop. Anz. 51 : 41-46.
- Drusini AG (1997) The coronal pulp cavity index: a biomarker for age determination in human adults. American J. of Physical Anthropology 103: 353-363.
- Drusini AG, Zadzinska E, Carrara N, 2000, The comparison between two age estimation methods based on human teeth. Przeglad Antropologiczny-Anthropological Review 63: 95-101.
- Grobskopf B (1990) Individualaltersbestimmung mit Hilfe von Zuwachsringen im Zement bodengelagerter menschlicher Zahne. Z. Rechtstmed. 103: 351-359.
- Gustafson G (1950) Age determinations on teeth. J. Am. Dent. Assoc. 41: 45-54.
- Haertig A, Durigon M (1978) Identification dentaire. Comparaison des méthodes de Gustafson et deShiro-Ito (modifiée). Bull.Méd.Lég. Toxicol. 21: 603-608.
- Harris EF, 2008, Statistical applications in dental anthropology. In: Irish J.D., Nelson G.C., 2008, Technique and Application in Dental Anthropology. Cambridge university Press, Cambridge, pp. 35-67.
- Helfman PM, Bada JL (1975) Aspartic acid racemisation in tooth enamel from living humans. Proc. Nat. Acad. Sci. U.S.A. 72: 2891-2894.
- Hillson S. (1986) Teeth. Cambridge: Cambridge University Press.
- Huda TFJ, and Bowman JE (1994) Variation in cross-striation number between striae in an archaeological population. Int. J. Osteoarchaeol. 4 : 49-52.
- Huda TFJ, and Bowman JE (1995) Age determination from dental microstructure in juveniles. Am. J. Phys. Anthropol. 97 : 135-150.
- Ikeda N, Umetsu K, Kashimura S, Suzuki T, and Oumi M (1985) Estimation of age from teeth with their soft X-ray findings. Jap. J. For. Med. 39 : 244-250.
- Ito S (1972) Research on age estimation based on teeth. Jap. J. Leg. Med. 26 : 31-41.
- Ito S (1975) Age estimation based on tooth crowns. Int. J. Forens. Dent. 3 : 9-14.
- Johanson G (1971) Age determination from human teeth. Odontologisk Revy 22 : 1-126.
- Kvaal S., and Solheim T. (1989) Fluorescence from dentin and cementum in human mandibular second premolars and its relation to age. Scand. J. Dent. Res. 97 : 131-138.
- Lamendin H, Cambray JC (1981) Etude de la translucidité et des canalicules dentinaires pour l’appréciacion de l’age. Med Leg.Droit Med. 24 :489-499.
- Lamendin H, Humbert JF, Tavernier JC, Brunel G, and Nossintchouk R (1990) Estimation d’âge par une méthode à deux critères dentaires. Le Chirurgien-dentiste de France 539: 93-96.
- Lamendin H, Baccino E, Humbert JF, Tavernier JC, Nossintchouk RM (1992) A simple technique for age estimation in adult corpses : the two criteria dental method. J. Forencic Sci. 37:1373-1379.
- Langland OE, Langlais RP, and Morris CR (1982) Principles and practice of panoramic radiology. Philadelphia: Saunders.
- Lipsinic FE, Paunovich E, Houston GD, and Robison S (1986) Correlation of age and incremental lines in the cementum of human teeth. J. Forensic Sci. 31: 982-989.
- Liversidge H., 2008, Dental age revisited. In: In: Irish J.D., Nelson G.C., 2008, Technique and Application in Dental Anthropology. Cambridge university Press, Cambridge, pp. 234-252.
- Lorton L (1988) Discussion on «Failure of use of cemental annulations in teeth to determine the age of humans. J. Forensic Sci. 33: 1305-1306.
- Lovejoy CO, Meindl RS, Mensforth RP, Barton TJ (1985) Multifactorial determination of skeletal age at death: a method and blind tests of its accuracy. Am. J. Phys. Anthropol. 68 : 1-14.
- Maples WR (1978) An improved technique using dental histology for estimation of adult age. J. Forensic Sci. 23 : 764-770.
- Masters PM (1986) Age at death determinations for autopsied remains based on aspartic acid racemization in tooth dentin: importance of postmortem conditions. Forensic Sci. Int. 32 : 179-184.
- Metzger Z, Buchner A, and Gorsky M (1980) Gustafson’s method for age determination from teeth. A modification for the use of dentists in identification teams. J. Forensic Sci. 25 : 742-749.
- Miles AEW (1958) The assessment of age from the dentition. Proc. Roy. Soc. Med. 51 : 1057-1060.
- Miles AEW (1963) The dentition in the assessment of individual age in skeletal material. In DR Brothwell (ed.): Dental Anthropology. London: Pergamon Press, pp. 191-209.
- Morse DR (1991) Age-related changes of the dental pulp complex and their relationship to systemic aging. Oral. Surg. Oral Med. Oral Pathol. 72 : 721-745.
- Nalbandian J, and Sognnaes RF, 1960, Structural age changes in human teeth. From: Ageing. Amer.Ass. Adv. Sci., pp. 367-382.
- Naylor JW, Miller WG, Stokes GN, and Stott GG (1985) Cemental annulation enhancement: a technique for age determination in man. Am. J. Phys. Anthropol. 68 : 197-200.
- Nowell GW (1978) An evaluation of the Miles method of ageing using the Tepe Hissar dental sample. Am. J. Phys. Anthropol. 49 : 261-276.
- Ogino T., and Ogiino H. (1988) Application to Forensic Odontology of aspartic acid racemization in unerupted and supernumerary teeth. J. Dent. Res., 67 : 1319-1322.
- Ogino T., Ogino H., and Nagy B. (1985) Application of aspartic acid racemization to Forensic Odontology: post mortem designation of age at death. Forensic Sci. Int. 29 : 259-267.
- Ohtani S, and Yamamoto K (1987) Age estimation using the racemization of aspartic acid on human dentin. Jap. J. Leg. Med. 41 : 181-190.
- Philippas GG (1961) Influence of occlusal wear and age on formation of dentine and size of pulp chamber. J. Dent. Res. 40 : 1186-1198.
- Philippas GG, and Applebaum E (1966) Age factor in secondary dentine formation. J. Dent. Res. 45 : 778-789.
- Philippas GG, and Applebaum E (1967) Age changes in the permanent upper lateral incisor. J. Dent. Res. 46 : 1002-1009.
- Philippas GG, and Applebaum E (1968) Age changes in the permanent upper canine teeth. J. Dent. Res. 47 : 411-417.
- Pilz W (1959) Das Transparenzphanomenon der Zahnwurzel als ein Ausdruck der Biomorphose des menschlichen Dentin. Z. Alterforsch. 13: 139-152.
- Pindborg JJ (1970) Pathology of Dental Hard Tissues. Copenhagen: Munksgaard.
- Poyton HG (1982) Oral Radiology. Baltimore/London: Williams & Wilkins, pp. 50-51.
- Ritz S., Schultz HW, Peper C (1993) Postmortem estimation of age at death based on aspartic acid racemization in dentin: Itsbility for root dentin. Int. J. Legal Med. 105: 289-293.
- Robinson HBG, and Boling LR (1952) Teeth and jaws. Problems of ageing. Baltimore: Cowdry, pp.447-480.
- Rosing FW, Kvaal S.I. (1998) Dental age in adults – a review of estimation methods. In: Dental Anthropology. Fundamentals, limits, and prospects. Springer Verlag, Wien, pp. 443-468.
- Schmidt C.W., 2008, Forensic dental anthropology: issues and guidelines. In: Irish J.D., Nelson G.C., 2008, Technique and Application in Dental Anthropology. Cambridge university Press, Cambridge, pp. 266-292.
- Smith BH (1984) Rates of molar wear: implications for developmental timing and demography in human evolution. Am. J. Phys. Anthropol. 63 : 220.
- Solheim T, and Sundnes PK (1980) Dental age estimation of Norwegian adults. A comparison of different methods. Forensic Sci. Int. 16 : 7-17.
- Solheim T (1992) Amount of secondary dentin as an indicator of age. Scand. J. Dent. Res. 100: 193-199.
- StottGG, Sis RF, Levy BM (1982) Cemental annulation as an age criterion in forensic dentistry. J. Dent. Res. 61: 814-817.
- Takei T, (1984) Age estimation from dental attrition and state of dental treatment. Nihon Univ. School Dent. 26: 119-132.
- Tomaru Y, Uchiyama Y, Kobayashi K, Mikami H, Endo M, Tsukamoto T, Terezawa K (1993) Age estimation from tooth attritios of lower incisors. Jpn.J.Legal Med. 47:13-17.
- Walker PL, Dean G, Shapiro P (1991)
- Wei B, Feng L (1984) A trinomial regression formula of estimation age with both the degree of tooth attrition and the tooth index. Acta Anthrop. Sinica3: 270-276.
Correspondence:
Dr. Andrea G. Drusini.
Dipartimento di Scienze Medico-Diagnostiche e Terapie Speciali,
Università di Padova, Via Giustiniani, 2- Padova
35128, Italia.